Exploring Cancer Treatment Strategies — Target Therapies
The global cancer burden is driving technological advances in cancer research and treatment. Currently, nearly nine million people a year lose their lives to cancer. This equates to approximately 22,000 cancer deaths per day [1]. This burden is expected to grow as the world’s population expands and ages. There is a clear urgency to finding better treatments for current and future cancer patients navigating through the phases of cancer care.
The number of new US Food and Drug Administration (FDA) approved therapies and new uses of cancer therapies have been increasing dramatically in recent years. Majority of these improvements are associated with enhancements in patient survival and/or quality of life. Over the next few weeks, we will explore the pertinent cancer care areas of targeted therapy [2], medical, surgical, and radiation oncology.
Advances in cancer care and research allow for precision, and targeted therapy is the foundation of precision medicine. Targeted therapy aims to treat cancer by targeting the changes in cancer cells that help them grow, divide, and spread. This cancer treatment uses drugs and is different from traditional chemotherapy. It works by targeting specific genes and proteins related to cancer growth and spread of tumors.
To better understand the “targets” of targeted therapy, it is beneficial to know how cancer cells grow. Cells are the building blocks of every tissue in our bodies. There are many different cell types with different, yet specific functions. Cancer starts when a cell has a mutation – certain genes of healthy cells change. Genes play the most critical role as they tell cells how to function. If genes are altered, so is the functionality of the cell, it might either divide abnormally or live too long. When cells grow out of control, tumors form.
As knowledge and findings about cellular level changes increases, cancer researchers are able to design intricate and promising therapies that target erroneous changes and ultimately block their effects. These aim to:
1) Block or turn off signals that tell cancer cells to grow and divide
2) Prevent cells from living longer than normal
3) Destroy the cancer cells
There are two main types of targeted therapy:
1) Small-molecule drugs: these drugs aim to block the process that helps cancer cells multiply and spread. An example of this type of targeted therapy is angiogenesis inhibitors. These drugs keep the tissue around the tumor from making blood vessels. Hence, anti-angiogenesis therapies starve the tumor by keeping new blood vessels from forming.
2) Monoclonal antibodies: these drugs block a specific target on the outside of cancer cells. They can also send toxic substances directly to cancer cells.
It is important to remember that not all cancers type are the same, and not all tumors have the same targets. Therefore, targeted therapies must match the mutation. For instance, if an individual has colorectal cancer, they are tested for the KRAS mutation. This mutation controls the growth and spread of a tumor, and about 40% of colorectal cancers have this gene. When this mutation is present, some targeted therapies are not effective. This identification helps the cancer care team provide the most effective treatment, and protects the patient from unnecessary side effects or treatment costs. With advances such as targeted therapy, healthcare professionals have the ability to match every tumor to the best possible treatment.
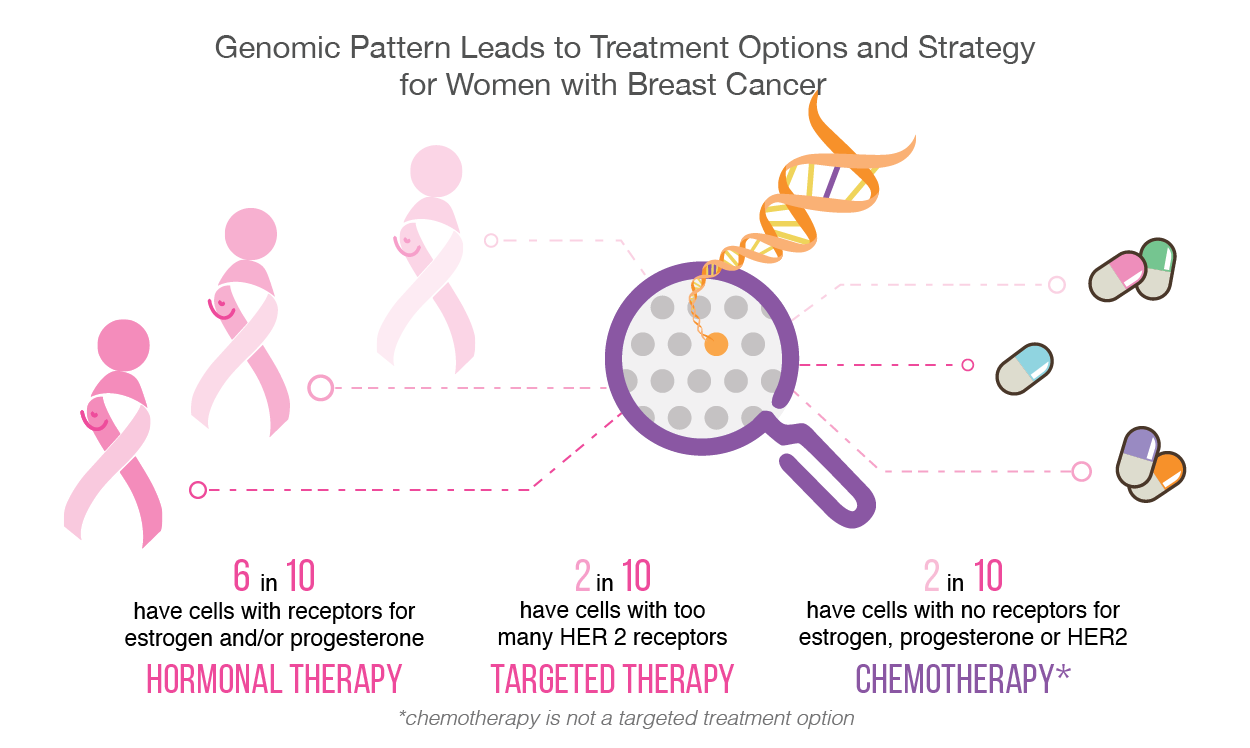
In the case of breast cancer, approximately 25% of all cases have too much of a protein called human epidermal growth factor receptor 2 (HER2). This protein induces tumor cell growth [3]. Fortunately, if the type of breast cancer is HER2 positive, then several targeted therapies are available to stop and limit its functionality. HER2-targeted therapies include Trastumab, Pertuzumab, and Neratinib. Some are taken in combination with one another, or along with chemotherapy.
Nearly all novel interventions have their own set of limitations and challenges, and targeted cancer therapies are no exception. One limitation is that cancer cells may become resistant to the therapy, meaning that this method will no longer be effective. This may happen if the target changes itself through mutation, or if the tumor finds a new alternative way to induce tumor growth. It is because of this limitation that most targeted therapies may work best in combination with multiple targeted therapies, and/or in combination with one or more chemotherapy drugs. A challenge is that drugs for some identified targets are difficult to develop as the target’s structure and its pathway are regulated within the cell and are not easily accessible.
One expected goal for targeted therapies is for them to be less toxic than traditional chemotherapy drugs. However, they too can have side effects that need to be tracked by healthcare professionals. Patients may easily assist in reporting their symptoms through patient-reported toxicity questionnaires. This sharing of knowledge and information will further assist the refinement and enhancement of targeted therapies.
Considering these identified obstacles, targeted therapies remain a novel and growing approach to curtailing cancer progression. Cancer care and research are no longer approached through a one-size-fits-all model. Care and treatment have become much more personalized. Doctors and the cancer care team now know that one cancer, whether it be colorectal or breast, can come in many different genetic types. Targeted therapy through the use of drugs and other substances has the ability to more precisely identify and attack cancer cells, providing a promising conduit to diminish or eliminate the cancer care journey altogether.
Written by Dorri Mahdaviani , who holds a Masters of Public Health (MPH) from the University of British Columbia (UBC). Her academic and professional interests include the areas of chronic illnesses, health care systems and childhood health and development.
Infographics and Design by Ann Wong, who holds a PhD in Biochemistry and Molecular Biology from the Faculty of Medicine at the University of British Columbia (UBC), Canada. She is an author of over 10 SCI publications, having taught at UBC and the Peking University Health Science Center (PUHSC) in Beijing